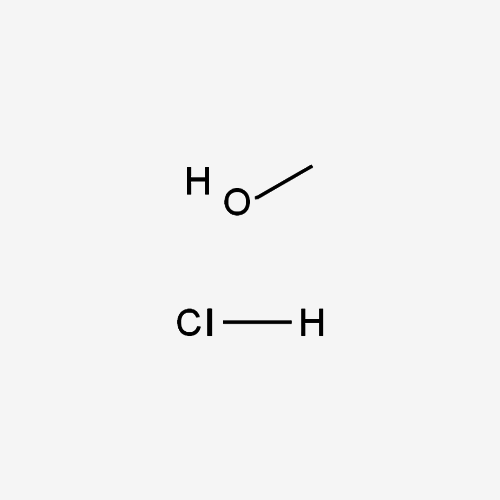
Understanding Methyl Alcohol HCL (Methanol Hydrochloride):
What is Methyl Alcohol HCL? Methyl Alcohol HCL, also known as Methanol Hydrochloride, is a chemical compound resulting from the reaction between methanol (CH3OH) and hydrogen chloride (HCl). Its molecular formula is CH3OH HCl. This clear, colorless liquid possesses unique properties that make it invaluable in various industrial applications.
Formula:
CH5ClO
Synonyms:
- Hydrochloride methanol
- HCl-methanol
- HCl methanol
- Methanol HCl
- Methanol-HCl
Properties of Methyl Alcohol HCL:
- Chemical Structure:Methyl Alcohol HCL comprises methanol molecules bonded to hydrogen chloride, offering distinctive chemical characteristics.
- Physical State:Typically, Methyl Alcohol HCL exists in liquid form at room temperature, with a boiling point approximately around 64.7°C.
- Reactivity:With its chemical composition, Methyl Alcohol HCL exhibits reactivity suitable for specific chemical synthesis processes.
- Solubility:It is soluble in water and many organic solvents, enhancing its applicability in various solutions and formulations.
Applications of Methyl Alcohol HCL:
Methyl Alcohol HCL serves as a crucial intermediate in organic synthesis, facilitating the production of pharmaceuticals, pesticides, and other fine chemicals.
Researchers utilize Methyl Alcohol HCL as a reagent in various experimental procedures, owing to its reactivity and solubility properties.
In certain formulations, Methyl Alcohol HCL acts as an additive in fuels, contributing to performance enhancement and stability.
Its precise composition makes Methyl Alcohol HCL valuable in analytical chemistry applications, particularly in chromatography techniques.
Benefits of Ammonium Formate:
Purity and Precision
Methyl Alcohol HCL offers high purity levels, ensuring consistency and reliability in synthesis processes.
Versatility
Its broad spectrum of applications underscores its importance across diverse industries, catering to different production needs.
Efficiency
As a chemical intermediate, Methyl Alcohol HCL streamlines synthesis processes, enhancing overall production efficiency.
Analytical Accuracy
Its precise composition aids in accurate measurements and analyses, particularly in analytical chemistry methodologies.
Safety Considerations:




While Methyl Alcohol HCL is valuable in various applications, safety precautions are paramount:
Adequate ventilation is necessary during handling to prevent inhalation of vapors.
Personal protective equipment such as gloves, goggles, and lab coats should be worn to minimize direct contact.
Proper storage in tightly sealed containers away from incompatible substances is essential to prevent accidents.
Care should be taken to avoid spills, as Methyl Alcohol HCL can be harmful upon skin contact or ingestion.
Proper disposal procedures should be followed to ensure environmental safety and compliance with regulations.
Unlocking Potential with Methyl Alcohol HCL: Methyl Alcohol HCL stands as a cornerstone in chemical synthesis, research, and various industrial processes. Its precision, versatility, and efficiency make it indispensable across multiple sectors. However, prudent handling and adherence to safety protocols are imperative to harness its benefits effectively. Trust in the reliability of Methyl Alcohol HCL to propel your endeavors towards success.