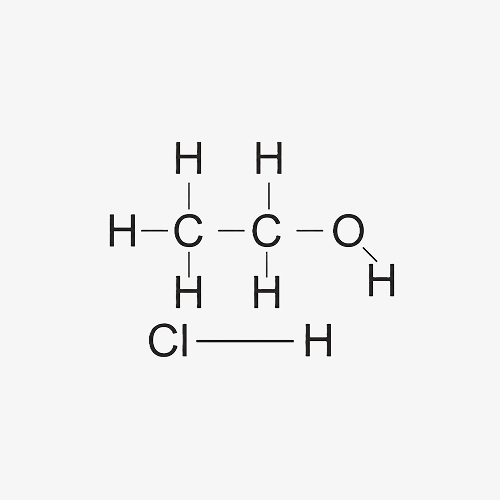
Hydrogen Chloride (HCI) in Ethanol (ETOH)
Description
Hydrogen Chloride (HCl) in Ethanol (EtOH) is a solution formed by dissolving hydrogen chloride gas in ethanol, a widely used alcohol solvent. This solution acts as a powerful acid catalyst in organic synthesis and is frequently employed in esterification, alkylation, and other acid-catalyzed reactions. The combination of HCl and ethanol provides a versatile reagent that is highly effective in both laboratory and industrial processes.




Category
Gas to Liquid Technology - Acids
CAS no
7647-01-0, 64-17-5
Purity
>=99%
Formula
C2H50H.HCL
Synonyms
Ethanol - Hydrochloride
Form
Liquid
Uses
• Anhydrous HCl source for moisture-sensitive reactions
• Salt formation of amines (amine → hydrochloride salts)
• Esterification catalyst in organic synthesis
• Deprotection / cleavage of acid-labile groups
• Activation of alcohols for substitution reactions
• Used in API & fine chemical synthesis as a controlled acidic medium