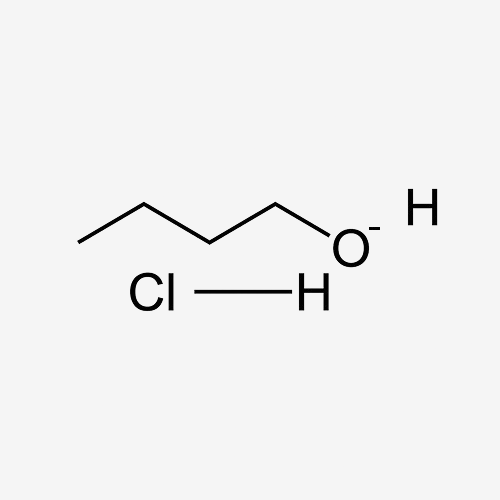
Hydrogen Chloride (HCL) in Butanol (BTOH)
Description
Hydrogen Chloride (HCl) in Butanol (BuOH) is a solution made by dissolving hydrogen chloride gas in butanol, an alcohol known for its moderate volatility and solvent properties. This solution serves as a strong acid catalyst and reagent, frequently used in organic synthesis for esterification, alkylation, and other acid-catalyzed processes. The combination of HCl and BuOH creates a reactive medium, ideal for various chemical transformations.




Category
Gas to Liquid Technology - Acids
CAS no
7647-01-0, 71-36-3
Formula
C4H100.HCL
Synonyms
Butanol - Hydrochloride
Form
Liquid
Uses
• Catalyst for esterification reactions (e.g., making butyl esters)
• Alcoholic HCl reagent for organic synthesis
• Cleavage of protecting groups in organic chemistry
• Salt formation of amines (HCl salts) in non-aqueous medium
• Used in pharmaceutical and fine chemical synthesis